Introduction.
Selecting the best available stem cell donor is crucial to optimize outcome after allogeneic hematopoietic cell transplantation (alloHCT). For several decades, matched sibling donors (MSD) were considered to be the best choice for all patients. Recently published data suggest that young unrelated donors (UD) may represent a favorable alternative. However, analyses in contemporary disease-specific registry cohorts are still rare. Therefore, we set out to investigate whether young UD provide a benefit for older patients with myeloid malignancies compared to MSD.
Patients and Methods.
We performed a retrospective study with data of the Deutsches Register Stammzelltransplantation (DRST) on patients 50 years and older who received a first alloHCT for AML, MDS, MDS/MPN or CMML between January 2010 and December 2020. We compared results of patients who were transplanted from MSD aged 50 years or older and young UD aged 35 years or younger who were HLA-compatible for HLA-A, -B, -C, and -DRB1. We evaluated the primary endpoint, event-free survival (EFS), and the secondary endpoints overall survival (OS), cumulative incidence of relapse/progression (CIR), non-relapse mortality (NRM), acute and chronic Graft-versus-Host Disease (GvHD) in multivariable Cox regression models.
Results.
In total, we analyzed data from 3460 patients. Median patient age was 63.5 years for 2225 patients with UD compared to 60.9 years for 1235 patients with MSD (p<.001). The median donor age was 27 years (range 18-35 years) for UD compared to 58 years (range 50-79 years) for MSD (p<.001). Overall, male donors accounted for 77.9% of UD compared to 49.3% of MSD (p<.001). Fewer male patients had female UD than female MSD (8.6% versus 30%, p<.001). Also, more cytomegalovirus (CMV) seronegative patients had CMV seronegative UD (34.9% versus 23%, p<.001). The percentage of alloHCT with UD increased in recent years, from 46% in 2010 to 72% in 2020 (p<.001). HLA-DQB1-mismatches were reported for 2.5% of UD. HLA-DPB1 matching information was available for 33.6% of UD.
We found no differences with respect to the distribution of Karnofsky performance status, disease risk, reduced intensity conditioning (circa 80% of patients) and graft source (almost exclusively PBSC) between the two donor types. GVHD prophylaxis differed significantly, more patients with UD received ATG compared to patients with MSD (87.4% versus 52.9%, p<.001).
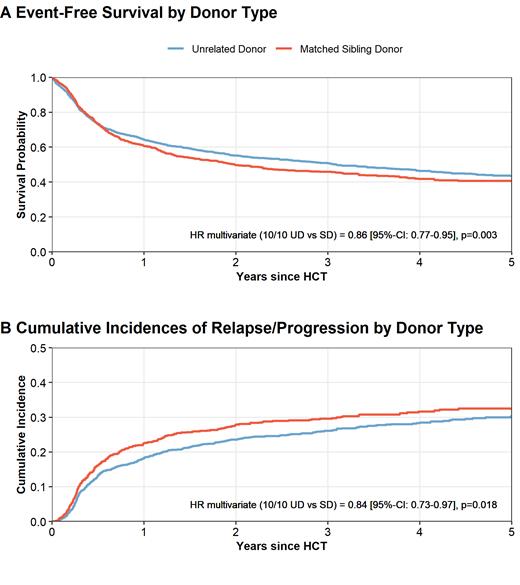
In univariate comparisons, we observed a trend towards improved EFS (HR 0.91, p=.07; Figure Panel A) and improved OS (HR 0.88, p=.03) for UD compared to MSD transplants. CIR was lower with UD (HR 0.87, p=.04, Figure Panel B) and NRM was not statistically different (HR 0.96, p=.6). The 150-days cumulative incidence of aGVHD was 28.2% for UD versus 31.3% for MSD transplants (p=.09). Corresponding numbers for the 1-year cumulative incidence of cGVHD were 31.3% and 39.4% for UD versus MSD transplants (p=<.001).
After multivariable adjustment for patient age, performance status, disease risk, conditioning type, stem cell source, and HLA-DQB1-mismatches, EFS and OS were significantly better for 10/10 UD compared to MSD transplantation with hazard ratios (HR) of 0.86 for EFS (p=.003) and 0.82 for OS (p<.001). After UD alloHCT we found a lower risk of relapse (HR 0.84, p=.018), less chronic GVHD (HR 0.7, p<.001) and a trend towards lower NRM (HR 0.87, p=.07).
Conclusions.
Selecting HLA-compatible UD over older MSD improves the chances of survival (both OS and EFS) after alloHCT for older patients with myeloid malignancies. This beneficial effect was mediated mainly by a reduced risk for relapse/progression suggesting more potent Graft-versus-Leukemia effects after UD transplantation despite less chronic GVHD.
Disclosures
Schetelig:BMS: Consultancy, Honoraria; Eurocept: Honoraria; Novartis: Honoraria; AstraZeneca: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; BeiGene: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria. Bug:Neovii: Other: Travel Grant; Pfizer: Honoraria; Gilead: Honoraria, Other: Travel Grant; BMS: Honoraria; Jazz: Honoraria, Other: Travel Grant; Novartis: Honoraria. Mueller:Gilead: Consultancy, Honoraria; Pfizer: Consultancy; Novartis: Consultancy; Amgen: Consultancy, Research Funding; Jazz: Honoraria. Stelljes:Novartis: Speakers Bureau; MSD: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Jazz: Speakers Bureau; abbvie: Speakers Bureau; Pfizer: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; medac: Other: Editorial and statistical support , Speakers Bureau; Amgen: Membership on an entity's Board of Directors or advisory committees; Gilead: Speakers Bureau. Middeke:Novartis: Honoraria, Research Funding.